2012 Annual Report
2012 Annual ReportTurning Stem Cells into Cures |
|||||||
President’s letter
|
Chair’s letter
|
||||||
Progress Toward TherapiesIn the past year, we’ve increased the number of projects headed toward patients by 80%.
|
We’ve invested more than $600 million in 77 projects moving toward therapies for 31 currently incurable diseases.
|
||||||
News From CIRM: 2012 in reviewA principal goal going forward is to connect our groundbreaking scientists with biotechnology and Pharmaceutical companies, which could provide additional funds and ultimately bring cellular therapies to market.
Jonathan Thomas, CIRM board chair Three projects in clinical trialsAt the end of 2011, we had 14 projects attempting to reach clinical trials. One year later, we have 18 projects at that stage, and 10 projects aiming to complete a trial. Three of those are already enrolling patients. Read about our therapy development projects Scientific progresThis year alone CIRM grantees published more than 150 papers describing their scientific progress. Stay up to date on these advances by reading our blog, or following us on either Facebook or Twitter. Institute of Medicine ReviewThe Institute of Medicine reviewed CIRM’s progress and called the agency a “bold social innovation.” We held a workshop to address their recommendations. Read our response New Strategic PlanOur new strategic plan refines how we expect to acheive our primary goal of bringing new therapies to patients. Read more about the plan Training the next generationCIRM has programs to bring students at all levels into stem cell science careers. These students will be tomorrow’s stem cell scientists, continuing Caliornia’s leadership in developing therapies for incurable disease and injury. Read about our training programs Global partnershipsCIRM’s partnerships with governmental funding agencies and institutions have leveraged more than $75 million for research toward stem cell-based cures. With partners working around the world the sun never sets on CIRM teams. Read more about our partners New research space opensAll 12 CIRM-funded lab spaces are now open, bringing together stem cell researchers and speeding discovery. Our $271M investment leveraged more than $800M in public and private support, and created 25,000 job-years for the state. Read about these facilities |
|||||||

CIRM congratulates
Dr. Shinya Yamanaka on his 2012 Nobel Prize for advances in stem cell biology |
|||||||
Funding Commitments |
|||||||
Total Funding:$1.8M |
Total Grants:586 |
Institutions Funded:60 |
Researchers Funded:478 |
||||
Funding in 2012:$307M |
Grants in 2012:54 |
Therapy projects77 |
Clinical trials:3 |
||||
Where our funding goes |
Disease Category |
“Our Programs train California’s diverse young people to build the state’s stem cell workforce and develop the next generation of new therapies”Bertram Lubin, President and Chief Executive Officer of Children’s Hospital & Research Center Oakland, CIRM Board Member Read about our training programs |
|||||
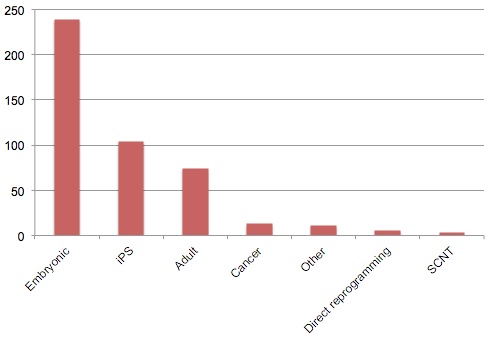 |
Stem Cell Type Funded |
||||||
Stories of HopeThroughout the year we hear from people who have benefited from stem cell therapies or who are still waiting for cures. These are their stories.
|
|||||||
CIRM Financials |
 |
 |
|||||








